|
Then, half of that amount in turn decays in the following half-life. After one half-life passes, half of the remaining nuclei will decay in the next half-life. 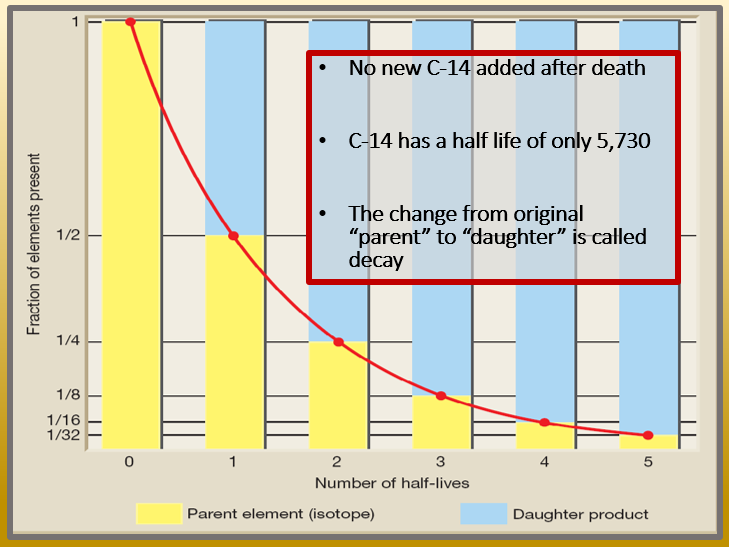
The time in which half of the original number of nuclei decay is defined as the half-life, t 1 2 t 1 2. Why do we use the term like half-life rather than lifetime? The answer can be found by examining Figure 22.27, which shows how the number of radioactive nuclei in a sample decreases with time. Here we will explore half-life and activity, the quantitative terms for lifetime and rate of decay. That means they have shorter lifetimes, producing a greater rate of decay. For example, radium and polonium, discovered by Marie and Pierre Curie, decay faster than uranium. However, some nuclides decay faster than others. 
Half-Life and the Rate of Radioactive Decay
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
